A New aMednovus Unveils New MRI Safety Detector: First Pass-Through With Introductory Pricing Below $10,000
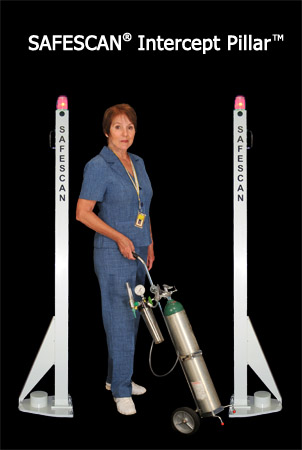
Leucadia, CA, April 30, 2009 — Mednovus, Inc., announced the release of their new SAFESCAN® INTERCEPT PILLAR™ ferromagnetic detection system this week at the Magnetic Resonance Manager’s Society (MRMS) annual meeting in Tampa, Florida. The product promises to help providers improve MRI safety for patients and staff.
The new system is comprised of a pair of coordinated independent pillars used to help identify the presence of ferromagnetic metals on persons or in objects in order to help keep them from entering the enormously powerful magnetic fields generated by magnetic resonance imaging (MRI) equipment.
 Why Ferromagnetic Detection?
Why Ferromagnetic Detection?
Thousands of times every year potentially dangerous magnetic materials are brought into MRI rooms. The profound attractive force that the MRI’s magnetic field (often up to 60,000 times the Earth’s own magnetic field) can twist or propel many of these objects with lethal force.
Conventional metal detectors can not effectively discriminate between non-magnetic metals used safely in the MRI environment (such as aluminum or titanium) and common – potentially lethal – ferromagnetic materials (such as iron, nickel or cobalt). These “airport style” metal detectors are specifically recommended against for pre-MRI screening, while ferromagnetic-only detectors have been explicitly endorsed by multiple best-practice authorities, including the American College of Radiology, the Department of Veterans Affairs, the UK’s MHRA.
Why the Intercept Pillar? Flexibility.
The Intercept Pillar allows MRI providers adhering to the best-practice calls for ferromagnetic pre-screening to have an unprecedented level of flexibility with a SAFESCAN product.
“Until they’ve used a ferromagnetic detector, some providers must make their best guess at how the technology will most effectively integrate with their operations, “ offered Tobias Gilk, President and MRI Safety Director of the Mednovus SAFESCAN company. “And sometimes the feedback that these instruments provide leads providers to make significant changes to their operational procedures. This new Intercept Pillar product affords MRI centers the flexibility for repositioning, reconfiguration and wholly different sets of capabilities, with a single product.”
Because the system is composed of two independent, freestanding detectors, users can have them positioned at distances that best work with their needs and their suite layouts. And since the pillars are each lightweight and require only a wall outlet, it takes only a few minutes to relocate, reposition or reconfigure the system.
“We have provided fixed-width portals, and these are excellent products for many of our customers,” offered Gilk. “But a good number of providers want to make decisions that afford them greater flexibility, whether it’s for when their new magnet arrives, or for the time they’re needing an interim trailer, or for their planned addition. The Intercept Pillar does exactly that.”
Why the Intercept Pillar? Cost-Effectiveness.
Mednovus and its partners have collaborated on the designs of dozens of ferromagnetic detection systems and continue to refine sensitivity and optimize the cost-effectiveness of their products. The new Intercept Pillar is the first pre-MRI pass-through ferromagnetic detector initially marketed for $9,500.
“This is an introductory price, to be sure, but we wanted this product to be affordable to providers who have wanted – and have been waiting for – ferromagnetic detection but felt that, budgetarily, it had been out of their reach,” said Gilk. “We know that this is a revolutionary product, at a revolutionary price.”
As Mednovus is unveiling the new addition to the SAFESCAN product family, the company is manufacturing an unprecedented number of pre-ordered units. “This Pillar system is clearly a product that the MRI community has needed,” stated Gilk. “We’ve never had these numbers of pre-orders for an unreleased product before, and we’re diligently working to make certain that production timetables aren’t stretched for the Intercept Pillar or our other SAFESCAN products.”
Mednovus, Inc. is the performance leader in projectile-screening instruments for accident and loss reduction in MRI suites. Their patented SAFESCAN® technologies include the walk-through Intercept Pillar™ System and the hand-held Target Scanner™. Mednovus also offers consulting services.
