When Patient Safety Depends on Data: How AI Is Reshaping DSCSA Compliance
By Upender Solanki
Patient safety has always been the central promise of the pharmaceutical supply chain. In the United States, that promise is enforced through the Drug Supply Chain Security Act (DSCSA), a federal law enacted in November 2013 as part of the Drug Quality and Security Act (DQSA) to prevent counterfeit, diverted, or otherwise unsafe drugs from entering the supply chain. The law mandates serialized, electronic, and interoperable traceability for prescription drugs at the saleable unit level.
Under DSCSA, every prescription drug must be uniquely identified, traced, verified, and exchanged electronically across a complex ecosystem that includes manufacturers, repackagers, wholesale distributors, dispensers such as pharmacies and hospitals, and third-party logistics providers (3PLs). The intent is straightforward: To ensure that every medication reaching a patient is legitimate, safe, and traceable.
In practice, this has proven far more difficult. More than a decade after the law was passed, many pharmaceutical supply-chain stakeholders continue to struggle with operational execution, data synchronization, and real-time exception handling. These challenges are no longer theoretical abstractions. They affect drug availability, shipment acceptance, and ultimately patient care.
Artificial intelligence, applied responsibly and in alignment with regulatory standards, offers a path to addressing these challenges in a more durable way.
DSCSA execution at scale: Why compliance becomes fragile
DSCSA compliance is rarely undermined by a single system failure. More often, it breaks down across handoffs—between systems, partners, and processes that were never designed to operate as a coordinated whole.
Most pharmaceutical organizations rely on a fragmented technology landscape that includes packaging line and site servers, ERP platforms, warehouse management systems, serialization repositories, middleware, and external networks used to exchange serialized data with trading partners. Each component may function as intended in isolation. The difficulty emerges when serialized processes must execute seamlessly across all of them.
Consider a common scenario: A manufacturer shipping serialized product to a wholesaler. Unique serial numbers are generated centrally, provisioned to packaging lines, printed and verified on saleable units, and reported back to enterprise traceability systems. Units are aggregated into cases and pallets, with parent-child relationships captured digitally. The physical aggregation on the warehouse floor and the corresponding digital hierarchy must remain perfectly aligned.
From there, serialized products move from packaging lines to warehouse locations—often across geographies and, in some cases, across national borders, particularly when contract manufacturing organizations are involved or packaging operations span multiple sites. As outbound shipments are prepared, the exact serialized units being shipped are scanned, verified, and associated with shipping containers. Serialization data covering serial numbers, aggregation, locations, and timestamps must be transmitted electronically to the trading partner before, or at least alongside, the physical shipment.
At receipt, the wholesaler validates the product against the received data. If information is missing, delayed, or incorrect, the shipment cannot be accepted.
Even when the medication itself is fully legitimate and urgently needed, a failure at any step can lead to quarantine, rejection, or return. Data issues quickly translate into operational disruption, with downstream consequences for patients.
When this complexity extends across the broader ecosystem—adding CMOs, 3PLs, secondary wholesalers, dispensers, returns, repackaging, inter-warehouse transfers, and drop shipments—the fragility of serialized execution becomes impossible to ignore.
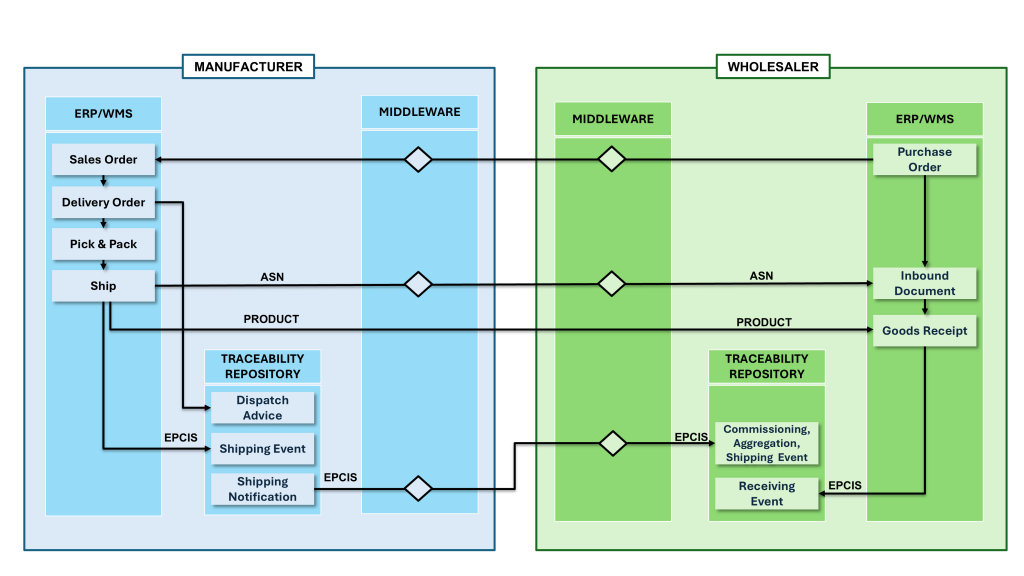
Figure 1. Serialized outbound shipment data exchanges where timing, synchronization, and EPCIS completeness determine downstream acceptance under DSCSA.
Why traditional monitoring falls short
From a technical perspective, a single DSCSA inbound or outbound flow may involve 10 or more potential failure points across systems and partners. Common issues include delayed EPCIS messages, master data mismatches, broken system handoffs, middleware transmission failures, and incorrect aggregation hierarchies.
In many organizations, these failures are detected only after a shipment arrives and cannot be accepted. Monitoring tends to focus on individual system transactions instead of end-to-end process completion. Teams compensate with manual checks, custom scripts, after-the-fact reconciliation, or large support groups responding to escalations once the damage is already done.
This reactive model carries significant cost and risk. More importantly, it creates conditions where life-saving drugs are delayed for reasons unrelated to manufacturing or logistics capacity. Data reliability becomes the limiting factor.
These weaknesses became especially visible under conditions of extreme stress.
When data reliability became mission-critical
During the COVID-19 pandemic, pharmaceutical supply chains faced unprecedented pressure. High-volume, time-sensitive distribution of vaccines and therapeutics left no tolerance for serialization or traceability failures. Shipments could not wait in quarantine for missing data to be reconciled, and downstream partners could not rely on assumptions when accepting product.
In advising and supporting DSCSA-regulated distribution during this period, one lesson became unmistakably clear: compliance controls designed to protect patients must also function reliably under real-world conditions, including surges in volume, compressed timelines, and complex partner coordination. When data failed to arrive on time, the consequences were immediate and tangible.
Those experiences fundamentally shaped how I think about DSCSA execution today. They exposed the limits of traditional monitoring approaches and reinforced the need for systems that anticipate failure, not just report it.
Designing an intelligent compliance layer
Building resilient DSCSA compliance requires a shift from monitoring transactions to understanding processes as they unfold across systems and partners.
Through the design and deployment of AI-enabled compliance monitoring solutions across serialized supply chains, several core principles have taken shape. First, success cannot be inferred from a single system event. A posted goods issue or an advanced shipping notice does not guarantee that all required serialization data has been generated, transmitted, received, and validated downstream. Compliance must be assessed at the process level, across the full chain of custody.
This begins with defining meaningful process milestones—such as the expected receipt of EPCIS data within a specific time window relative to shipment creation or arrival—and continuously validating those expectations against actual execution. When deviations occur, intelligent systems must determine why, not simply flag that something went wrong.
AI agents trained on GS1 EPCIS standards enable this level of contextual understanding. They can distinguish between timing issues, structural errors, master data discrepancies, and sequencing problems, providing precise insight into root causes. That insight supports targeted corrective action instead of generic alerts that overwhelm operations teams.
Equally important is orchestration. Intelligent compliance does not stop at detection. Systems must initiate follow-up actions, trigger partner communication, open support tickets, or escalate issues before shipments are impacted. Over time, trend-based learning allows thresholds to adapt to real-world partner performance, improving signal quality while preserving regulatory rigor.
Human oversight remains essential. In regulated environments, AI must operate with transparency and governance, augmenting expert judgment rather than replacing it. Human-in-the-loop models ensure accountability while allowing automation to address scale and complexity.
A critical distinction in this approach is the difference between transaction-level confirmation and true process completion. Many legacy monitoring models assume success once a single system event posts successfully—for example, when a shipment is created or an advance shipping notice is sent. In practice, DSCSA compliance depends on a chain of interdependent events across systems and partners, any one of which can fail silently. Process-level intelligence focuses on whether that full sequence has executed as expected, end to end, providing a more accurate and operationally meaningful view of compliance than isolated transaction checks ever can.
What changes when compliance actually works
When DSCSA compliance is operating effectively, the difference is felt well beyond audit readiness or reporting accuracy. Issues surface earlier in the lifecycle, often while there is still time to intervene—before a shipment arrives at a receiving dock or a trading partner is forced to halt acceptance. That early visibility changes the nature of response, shifting teams from reactive escalation to deliberate resolution.
Over time, this has a compounding effect on operations. Exceptions are addressed closer to their source, resolution paths become more predictable, and the volume of manual monitoring begins to decline. Compliance, IT, and supply-chain teams spend less time chasing missing data and more time managing outcomes, while collaboration with trading partners improves as issues are identified and resolved with greater context and clarity.
The most meaningful shift, however, is confidence in the data itself. Serialized information stops being treated as a potential liability and becomes a reliable operational asset—one that downstream partners can trust and act on without hesitation. That reliability is what ultimately protects patient access, by allowing legitimate, urgently needed medications to move through the supply chain without unnecessary delay or disruption.
Patient safety is a data discipline
DSCSA has significantly strengthened the integrity of the pharmaceutical supply chain. Over the past decade, one operational reality has emerged across the pharmaceutical supply chain: Patient safety depends on data moving correctly, completely, and on time.
AI, used responsibly and transparently, allows organizations to uphold both the letter and the intent of DSCSA by ensuring that compliance controls function reliably at scale. This shift does not weaken regulatory safeguards. It makes them operationally sustainable.
The future of pharmaceutical compliance is serialized, intelligent, and adaptive. That future is already emerging across the industry.
Upender Solanki is Chief Executive Officer of Novatio Solutions, where he leads AI-enabled digital transformation initiatives across pharmaceutical supply chains, healthcare systems, and other highly regulated industries. With 20 years of global experience in Life Sciences, SAP supply chain architecture, serialization, and regulatory compliance, he has advised manufacturers, distributors, wholesalers, and healthcare organizations on large-scale DSCSA and traceability programs, including initiatives supporting compliant distribution during the COVID-19 pandemic.
Solanki has contributed directly to national and global serialization standards development through participation in the Healthcare Distribution Alliance (HDA) Verification Router Service (VRS) Task Force and multiple GS1 industry workgroups, including the Lightweight Messaging Standard, Certificate of Conformance (CoC), and Core Business Vocabulary (CBV) initiatives. These efforts helped shape the technical frameworks and interoperability standards that underpin DSCSA-compliant verification and traceability across the U.S. pharmaceutical supply chain. He has also presented at HDA and GS1 annual industry forums on serialization and compliance architecture. He holds a Bachelor of Engineering in Information Technology. Contact him at Upender.solanki@novatiosolutions.com.
