Common Cause Analysis
May / June 2010
![]()
Common Cause Analysis
A large health organization used common cause analysis to investigate medication errors throughout the system, identify solutions, and reduce adverse events in high-risk medications by 50%.
To improve medication safety, many healthcare systems implement a technology (such as barcode at point of care) or a best practice (such as double-check of high-risk medications). This approach turns performance improvement into experimentation with other people’s solutions for other people’s system problems — the assumption being all providers share the same system problems. To improve its medication process reliability, OhioHealth used a different approach — common cause analysis. Instead of applying other’s solutions for other’s system causes, OhioHealth identified causes of medication error in its own systems and then identified proven solutions for those causes. This approach resulted in a 50% reduction in medication safety events involving high-risk medications over 3 years.
According to the Institute of Medicine’s (IOM) 2000 report To Err Is Human, medication errors, whether occurring within the facility environment or in the community setting, cause at least 7,000 deaths per year. Medication errors and adverse drug events also cause significant morbidity — impacting many patients. Adverse events are defined as those instances where a patient is harmed as a result of medical care (not associated with the patient’s underlying condition). The largest source of errors involves medications, making efforts to identify, analyze, and prevent errors vitally import to patient safety.
Analysis of Significant Errors and Events
Serious medication errors or adverse drug events involving death or significant patient harm are analyzed through root cause analysis (RCA). Root cause analysis is a process used to identify underlying causes of errors and contributing factors related to the event and to design an action plan to prevent recurrence of the event(s). This process can also be performed on events that cause minimal harm to the patient (precursors), near miss events caught before they reach the patient, or on clusters of events.
While learning from one high-severity event is necessary and often uncovers process gaps, there are usually multiple acts that lead to harm and multiple system causes leading to those acts. This effect is clearly shown in James Reason’s Swiss Cheese Model, where active and latent (system) errors coincide to allow adverse events. Determining where to focus limited resources becomes a challenge. At times it is necessary to prioritize the approach to the vulnerabilities in our systems.
Common cause analysis (CCA) aggregates acts and causes from multiple events to identify the common causes of those events. While it is beneficial to identify root causes and process gaps for single events, using CCA to examine multiple events allows an organization to identify the depth and breadth of system vulnerabilities (Table 1). Aggregating all of the root cause analyses from an institution or system is one way to use common cause analysis.
| Root Cause Analysis | Common Cause Analysis |
| Single case or a few related cases | Many or all cases |
| Event directed (examines a single event or adverse trend of related events). | Time or trend directed (examines all cases in a time period). |
| Efficient for diagnosing process, protocol, and technology causes. | Efficient for diagnosing people, leadership, and environment of care causes. |
| Investigates cause-and-effect relationships directly. | Infers cause-and-effect relationships using existing analyses. |
| An effective program lowers rates of serious harm by 50% every 2 years. | An effective program lowers rates of serious patient harm by 50% every 2 years, with 10% the resource allocation of root cause analysis. |
Table 1. Comparison of Cause Analysis Methods
OhioHealth Case Study
OhioHealth (www.ohiohealth.com) is a nationally recognized, not-for-profit, charitable, healthcare organization serving and supported by the community. Based in Columbus, Ohio, OhioHealth is a family of 18 hospitals, 23 health and surgery centers, home-health providers, medical equipment and health service suppliers throughout a 40-county area. OhioHealth hospitals in central Ohio are Riverside Methodist Hospital, Grant Medical Center, Doctors Hospital, Grady Memorial Hospital and Dublin Methodist Hospital.
Not long after To Err Is Human was published (IOM, 2000), the OhioHealth medication safety steering committee reviewed various methods of identifying and reporting medication errors in an effort to consistently identify harm related to medication use (adverse drug event or ADE). Voluntary error reporting is subject to many factors that negatively impact its abilitiy to be considered a reliable measure over time. A more concrete metric was desired in order to gauge the success of concerted efforts to decrease medication-related harm within their facilities. It was clear that the overall desire was to decrease harm related to medication use. The goals of the group were threefold:
- To develop and adopt a method that would consistently identify ADEs, provide a valid way of measuring opportunity gaps, and measure the impact of systematic changes.
- To reduce preventable ADEs, thus providing safer care for our patients
- To provide a platform for comparison of processes aimed at reducing ADEs within the OhioHealth system.
|
|
ADE Trigger Tool and Chart Review
OhioHealth conducted a pilot study in several facilities using the adverse drug event (ADE) trigger tool (Rozich et al., 2003). The ADE trigger tool identifies patients who may have experienced an adverse drug event. Examples of triggers include orders for antidotes, abnormal laboratory values, or orders to abruptly stop a medication. The use of this tool provides a measure of harm to patients and moves away from the focus on error alone. This is important because not all errors result in harm to patients. In actuality, a small percentage of errors result in harm to patients.
The use of the ADE Trigger Tool begins with random chart selection to identify a group of charts for further review. This is generally 20 charts per month, with a 75-chart baseline review. A trained reviewer checks each chart for one or more of the 24 defined triggers. If a trigger is identified, the entire chart is then reviewed for details related to that trigger in an effort to determine whether the patient experienced harm related to an adverse drug event. Based on the results of the pilot, a modified ADE trigger tool was developed, which focused on three classes of high-risk medications: anticoagulants, insulin, and narcotics/sedatives. Seven triggers were chosen to support the identification of potential adverse drug events associated with the use of these high-risk drugs.
Each facility in the system then performed a 3-month baseline data collection using the modified ADE tool for each of the three categories of high-risk drugs including all of the identified triggers. This method differed from the Resar methodology (Rozich et al., 2003). A report at each institution was developed that identified all patients who had any one of the seven defined triggers, as opposed to random chart selection. This provided a more focused look at patients who were receiving known high-risk medications. Upon completion of the baseline data collection, each institution chose one of the three high-risk classes to focus their efforts for the upcoming year. Each facility then developed an action plan for reducing ADEs in their chosen medication focus. The goal was to reduce ADEs from the baseline by 10% over 12 months.
Development of Risk Reduction Strategies
The use of the trigger tool to identify an adverse drug event was only the first step in the project. While the reviewers were in the chart, they collected standardized data in an effort to identify trends in potential contributing factors. The data was collated to provide a mechanism for aggregating the causes. This allowed for the identification of trends, themes, and common causes of adverse events related to high-risk medication use. Once common causes were identified, evidence-based best practices (from within the system and/or in the primary literature) were incorporated into the action plans.
Results
In the first year of the project, the system demonstrated a 45% relative reduction in adverse drug events within the medication class each institution chose (Crea et al., 2004). This success was acknowledged in December 2004 with the first Award for Excellence in Medication Use Safety given by the American Society of Health-System Pharmacists Foundation (award funds provided by Cardinal Health) and a Best Practice Award from the American Society of Health-System Pharmacists.
In the second year of the project, each facility chose a second of the three high-risk medication classes to add to the initiative, and in the third and final year, all facilities were working to decrease adverse events within all three high-risk classes of medications. The results over the 3-year period remained stable, with the final overall reduction of approximately 50% across all three medication classes.
|
ASQ Webinar Root cause analysis is the most widely used quality tool in healthcare. A webinar is available on the American Society for Quality website that reviews the basic steps of root cause analysis and explores its history and future. To view this complimentary webinar, visit http://www.asq.org/webinars/root-cause-analysis-healthcare.html |
Common Causes and Risk Reduction Strategies
While each facility identified some slightly different causes, overall the identified causes tended to be very similar.
- Anticoagulation with Heparin and Warfarin
Dosing: Common causes identified with anticoagulation tended to be related to the dosing for both heparin and warfarin. Most institutions were utilizing a preprinted order for heparin dosing, but several smaller facilities implemented this standardized process.
Drug interactions: The most common cause of elevated INRs with warfarin therapy was the impact of drug interactions. While patients may have been stabilized at home, once they were admitted, addition of an antibiotic or other drug caused an elevation in the INR, requiring dosage alterations.
Concomitant anticoagulants and antiplatelet agents: Patients who were on multiple drugs impacting coagulation experienced an increased rate of bleeding events. Many patients admitted for myocardial infarction were on aspirin, heparin, Plavix, and may have received a glycoprotein IIb/IIIa receptor inhibitor or thrombolytic at an outlying facility. These patients demonstrated an increased risk for bleeding.
Risk reduction strategies: Comparing the number of events between facilities revealed that one facility had very few ADEs with anticoagulation, and when they did occur, they were of mild severity. It was determined that this facility had provided pharmacy dosing (inpatient and outpatient) for several years. Based on this apparent best practice, one of the other institutions implemented a similar pharmacy dosing service for anticoagulation (both heparin and warfarin) and now it is the common practice. Five years later, The Joint Commission added a National Patient Safety Goal intended to reduce the likelihood of patient harm associated with the use of anticoagulation and encouraged standardized practices around the dosing and monitoring of anticoagulation. - Hypoglycemia and Insulin
Knowledge of insulin pharmacokinetics and dosing: In general, sites identified the potential for confusion and lack of knowledge regarding the multiple insulin products and dosing strategies. The look-alike and sound-alike nature of these products contributes to the risk and strategies around floor stock were developed with this in mind.
Appropriate identification and treatment of hypoglycemia: While hypoglycemia is generally detected by serum glucose or fingerstick values, there seemed to be considerable variation in the way patients were treated for hypoglycemia.
Risk reduction strategies: Facilities that did not utilize preprinted and standardized sliding scales or basal/prandial orders identified this as an area for improvement. In addition, several institutions standardized, through the use of protocols, the treatment of hypoglycemia. The use of food, juice, glucose tablets and D50W were defined and the physician was notified of the hypoglycemic event. At least one institution completed a failure mode and effect analysis on the insulin administration process, and floor-stock insulin was minimized and standardized. Subsequent work around standardization of insulin pump processes and NPO orders has been completed. - Narcotics and Sedatives
There were several diverse issues identified with the use of narcotics and sedatives.
Patient-controlled analgesia (PCA): Several institutions identified inconsistencies with patient selection and dosing for PCA pumps (with or without continuous mode). Revisions to preprinted PCA orders and the use of pain control services were found in several action plans.
Epidural narcotic use: Most institutions utilized preprinted orders, however changes were made including standardization of the use of pulse oximetry (when, where, who, and type of monitoring), monitoring parameters such as respiratory rate, and when to administer reversal agents.
Range orders: Nurses tended to have variable interpretation and administration practices when range orders were written for narcotics. Several facilities worked to eliminate the use of range orders. The timing was coincidental again, as The Joint Commission was recommending the elimination of range orders. The use of an administrative physician within the pharmacy department for 3 days to discuss prescribing options with physicians proved to be very successful in changing practice.
Meperidine use: While most institutions had moved away from meperidine use for pain control, there still remained a few holdouts. This practice was discussed and altered after review and discussion of the risks with the use of this medication.
Hydromorphone dosing and administration: There was one commonality identified across the system with pain control: a lack of knowledge (among physicians, nurses, and others) related to the dosing of hydromorphone (Dilaudid). The common belief was that hydromorphone and morphine dosing was equivalent milligram for milligram. One institution implemented a very effective process: calling the physician when an order was written for a hydromorphone dose > 1 mg to inform them of the equivalent morphine dose and provide recommendations for dosing. More than 90% of doses were altered based on this immediate feedback.
Challenges and Lessons Learned
The results of this project were significant and very rewarding, but the project was not without challenges.
- The large-scale project warranted monthly communication. A subcommittee, including pharmacists, quality consultant nurses, and others was developed to provide a forum for assuring consistency across the system.
- Exclusion criteria. A manual was developed to assure that all sites were using the same exclusion criteria.
- Defining harm for all classes. Define what constitutes harm for each of the high-risk medications to assure consistency across the system to avoid subjectivity and provide inter-rater reliability.
- Return on investment (ROI). The benefit-cost ratio of the project was important in keeping leader commitment to making safety changes. The benefit calculations were based on the work of David Bates et al. (1997)—a savings of $4,700 for each preventable ADE, which is likely to be a conservative estimate.
- Identification of hypoglycemia patients. Initial methods of identifying patients who developed hypoglycemia related to insulin use were based on the pharmacy charge database. It was discovered that some institutions were only charging patients for insulin if they were using an insulin infusion or a non-formulary insulin. Thus the criteria for identifying these patients had to change. Additionally, when a low blood sugar was identified, it was difficult to find documentation related to the patient’s signs and symptoms as evidence of an adverse event.
- Benefit (and difficulties) of using a denominator. One institution demonstrated a relatively flat rate of ADEs with anticoagulants over a 1-year period. Upon further investigation and work with denominators, it was determined that the facility actually had a decrease in the incidence of events related to anticoagulation. The institution had recently started an open-heart program, and the number of patients receiving anticoagulation had increased significantly from baseline. Therefore, when the denominator was employed, an apparently steady rate of events turned into a reduction in events.
- Committing necessary resources such as staff and time required for chart review. The time required for chart review increased with the scope of the study. To avoid the addition of resources, the reviews were accomplished through sampling techniques similar to those defined by the Centers for Medicare and Medicaid Services (CMS) for reporting quality measures. In addition, the larger institutions utilized pharmacy residents to assist with the chart reviews.
A Comment on Approach
The OhioHealth common cause analysis is an excellent example of the third of three methods of common cause analysis:
- Stream Analysis
- Change (or Difference) Analysis
- Multi-dimensional Pareto Analysis
Common cause analysis is an aggregating analysis where several less-consequential cases are combined together as a single cause analysis. The Joint Commission allows for aggregating analysis for cases of medication error and patient falls. Aggregating datasets is always more efficient in preventing recurrence of events than single-event root cause analysis. Good root cause analysis programs reduce event rates for serious events of patient harm by 50% in 2 years. An aggregating analysis of all cases produces similar results, but with one-tenth of the appraisal resources required.
The number of cases to be aggregated determines the method of analysis. Typically, 10 cases or fewer are better done as a Stream Analysis. Datasets with 10 to 25 cases are better done as Change Analyses, and greater than 25 cases are better done as a Pareto analysis.
OhioHealth’s analysis was performed using the third method, the multi-dimensional Pareto analysis, which is the best approach for the large numbers of cases seen in medication safety. To perform a multi-dimensional Pareto analysis, each case is entered into a spreadsheet or database in six basic dimensions plus any demographic information or case information special to the subject of the analysis (Table 2).
| Six Basic Dimensions | Other Typical Dimensions |
|
|
Table 2. Data Dimensions for Common Cause Analysis Method 3
Once the dataset is complete, Pareto charts are used as a visual guide to the dataset and to identify Dr. Juran’s “vital few” from “the irrelevant many.” Common causes are identified by inspecting the dataset for the unique sign and symptom pattern of a common cause. A typical Pareto chart for the dimension of organization committing the error is shown in Figure 1. Review the dataset from the viewpoint of each dimension, and form common cause theories that explain the observed data.
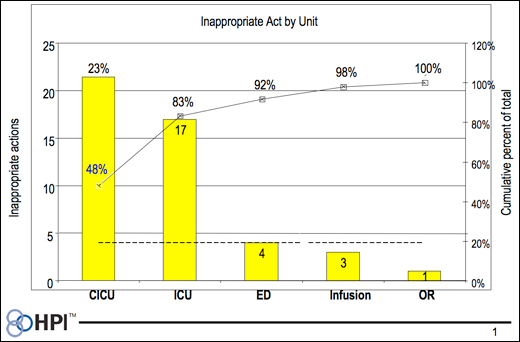
In practice, a dataset of 250 errors will yield around 25 theories. Each common cause theory is written in the structure of “what” happened “where,” with the “why” based on the observed system and human factors as symptoms. Each theory is very detailed and can appear to be very real, but only half of the theories are correct. Each common cause theory is then verified by study of data from sources independent of the common cause analysis dataset. If verified as correct, action is taken to remove the cause(s) from the system.
Recognizing a common cause theory embedded in the data is the value-added activity of the analysis. Common causes are identified by their signs and symptoms, much like diagnosing a disease. While common causes share several symptoms—as diseases share symptoms—each common cause has a unique symptom pattern. For example, a frequently observed pattern:
Complexity in protocol (system factor) coupled with user noncompliance (human factor) as a rule-based error.
This pairing could be symptomatic of: 1) excessive implementation requirements in the protocol, 2) poor compliance culture in the staff, and/or 3) low risk awareness by the user(s). The verification plan evaluates which of the three, if any, are present in the system and represent a sub-standard condition. If present and substandard, the system condition would require correction as a root cause.
Table 3 shows the last 2 years of common cause analysis results collected by one of the authors (Clapper) in his work with Healthcare Performance Improvement. These data were collected during the design phase of behavior-based patient safety culture interventions with U.S. healthcare systems. The safety behaviors are selected to prevent the human performance themes indicated by the data.
| System Causes (root cause theme) |
% acts | Human Performance (failure mechanism of care provider) |
% acts |
| Performance Culture | 53.8 | Critical Thinking | 33.2 |
| Process Design | 17.5 | Compliance | 22.5 |
| Structure / Function | 12.4 | Attention | 11.9 |
| Protocol Design | 11.5 | Communication | 9.5 |
| Technology / Environment | 4.9 | Knowledge (deficiency) | 8.1 |
| Normalized Deviance | 8.0 | ||
| Skill (deficiency) | 6.6 | ||
| 96 hospitals, 1429 cases, 4614 acts contributing to harm, 2008-2009 inclusive | |||
Table 3. Summary of Patient Harm Cases
In summary, diagnosing a common cause is not an easy task. Think of a root cause analysis case as a jigsaw puzzle in a brown paper bag. There are a few pieces missing to simulate gaps in information and a few pieces from other puzzles thrown in to simulate confounding information. Root cause analysis is still a straightforward task—literally piece together the information to see the “big picture.” Now think of common cause analysis as pieces from hundreds of jigsaw puzzles mixed together in a brown paper bag. Some common causes have several pieces and can be identified; many common causes have only one piece of data and may appear to belong to several possibilities or to none. The task is not easy, but the potential for results is enormous in that less-significant events can be used to improve patient care before an actual event of serious harm occurs.
References
Bates, D. W., Spell, N., Cullen, D. J., et al. (1997, January 22). The costs of adverse drug events in hospitalized patients. Adverse Drug Events Prevention Study Group. JAMA, 277(4), 307-311.
Crea, K. A., S. Herrin, T.P., Morehead, D., Snow, R. (2004). Reducing Adverse Drug Events Involving High-Risk Medications in Acute Care. Journal of Clinical Outcomes Management, 11(10), 640-646.
Institute of Medicine. (2000). To err is human: Building a safety health system. Washington, DC: National Academy Press.
Rozich, J. D., Haraden, C. R., Resar, R. K. (2003). Adverse drug event trigger tool: a practical methodology for measuring mediation related harm. Quality and Safety in Health Care, 12, 194-200.
Further Reading
Porras, J. (1994). Stream analysis. Reading, MA: Addison-Wesley.
Reason, J. (1997). Managing the risks of organizational accidents. Surrey, UK: Ashgate Publishing.
