Becoming a High-Reliability Organization Through Shared Learning of Safety Events
By Jeffrey Klenklen
Introduction
Nearly 20 years after the publication of the Institute of Medicine’s report To Err Is Human, healthcare providers look to methodologies used in other high-reliability organizations for transferrable strategies to reduce error in clinical care processes (Institute of Medicine, 2000). Organizations in industries such as nuclear power anticipate and mitigate errors by focusing on: 1) standard work through safe processes, 2) robust reporting, 3) simulation training, 4) establishing a safety culture, 5) rapid learning from events, and 6) robust process improvement (Birnhack et al., 2013).
Healthcare organizations often struggle to meaningfully translate these methodologies. This is complicated by the growing number of definitions and metrics for quality of care. Leaders struggle from a systematic perspective to define quality and safe care as they ruminate over this expanding plethora of metrics. Today’s successful leader must focus on safety, timely access, effectiveness, efficiency, equity, and patient-centeredness, while still producing enough income each year to finance improvements for the next (Ahluwalia, Damberg, Silverman, Motala, & Shekelle, 2017). Leaders are often forced to prioritize their focus using strategies from reliability science (Chassin & Loeb, 2013). Once focus is shifted away from an area of improvement, under the belief that reliability has been achieved, relapse can occur related to the processes that are no longer receiving focus.
Successful focus on and prevention of relapse requires leaders at all levels to constantly employ mindfulness through a concern over failure as a core strategy in maintaining reliability (Roberts, 1990). Organizations commit to resilience through embracing human-factor failures and rapidly learning from them when they occur (Carayon et al., 2014). Operationally, this constant concern over failure includes planned, focused, consistent shared learning from near-miss and actual events. This involves a clear understanding and acceptance that human factors such as noise, distraction, fatigue, and loss of situational awareness will disrupt staff in executing clinical processes (Carayon et al., 2014).
Whether at a single organization or at the system level, it is important to construct a shared learning model that can be successfully used to guide and maintain organizational mindfulness and learning about events. The core elements of this model include: 1) defining events that require rapid reporting, 2) rapid debriefing to detect ongoing or new latent-cause failures in the environment, 3) systems-based communication tools that focus on rapid communication of root causes of events and evidence-based solutions to prevent reoccurrence, 4) standardized methodology to communicate learning of these events to point-of-care staff across the organization(s), 5) aggregation of latent root causes of events that can be used for decision-based support development within the electronic health record (EHR) system and drive organization and system initiatives for failure prevention, and 6) provision of learning materials for each organization to be used in communicating about failures in events. Figure 1 provides a visual overview of the learning system model. Critical elements of the model will now be discussed.
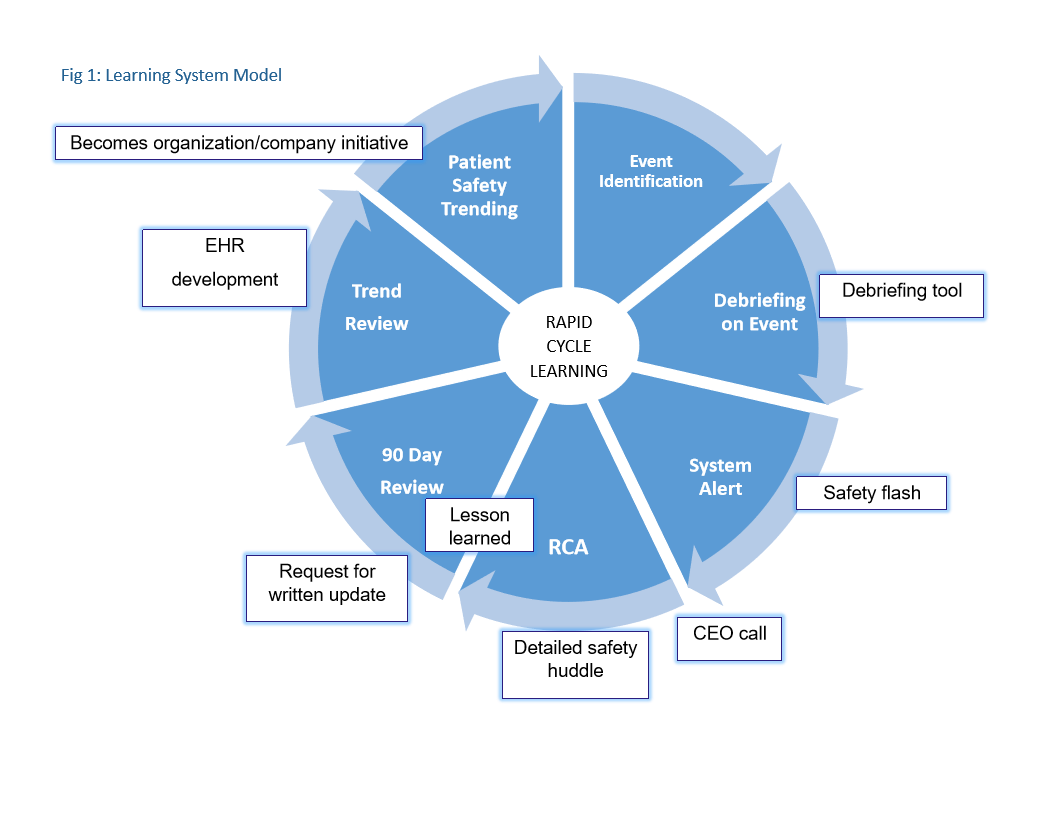
Defining serious safety events for debriefing and flash notification
It is important to identify events that require rapid debriefing and notification both within and external to the organization (Klenklen, 2017). Table 1 identifies events for rapid debriefing (Austin & Pronovost, 2015; The Joint Commission, n.d.). In debriefings, it is important to include near-miss events where safety practices have prevented patient harm.
Table 1: Events for Rapid Debriefing
Sentinel Events (As defined by the Joint Commission)
- Events resulting in major harm based on severity scoring
- For unexpected deaths report those that are related to potential medical error or unknown reason
- Near miss events that may result in patient(s) significant harm if recurrence
- Actual events that meet National Quality Forum (NQF) Serious Reportable Event Criteria
- Events involving potential allegation of patient abuse
- Death/disability to mothers/infants during hospitalization for delivery
- Fires in direct patient care areas
- Death in restraints meeting CMS reporting criteria
- Drug Diversion (patient safety events that impact patient care)
Organization debriefing with the care team is initiated no later than 24–48 hours after the identified event (see Figure 2).


A standardized team debriefing tool is used to document the debrief and detail next steps by the facility-based risk manager and/or patient safety officer. This process provides a framework for rapid decision-making if disclosure related to a medical error will be made to the patient and/or family. The debrief also serves as a summary of the critical elements from the event and can be used to send out key learnings across the local organization or system, whether in an email from leadership or another standardized communication methodology.
A safety flash using SBAR methodology can be issued if the event requires rapid communication across the enterprise to prevent reoccurrence (see Figure 3) (Haig, Sutton, & Whittington, 2006). This part of the rapid learning model can also be used to identify staff that make good catches using safety practices to prevent events. Staff are recognized both within the organization and at the system level to promote use of safety practices.

Learning from root causes of events
Analysis of the event identifies both proximate and, more importantly, latent event causes. This systems approach to event analysis acknowledges that human beings are fallible and holds that human factors are present and should be planned for. An error is seen as a consequence rather than a cause, having roots in the organizational systems that failed to prevent it (Karl & Karl, 2012).
At the organization level, action plans focus on responding to both proximate and latent event causes (Karl & Karl, 2012). Latent root causes are aggregated, and common human-factor failures are identified. A safety huddle is constructed to focus on evidence-based solutions and to help the organization or health system learn from the latent causes of these events (see Figure 3). The huddles utilize a standardized format that outlines 1) the background of the problem, 2) what the organization needs to know about the issue being discussed, and 3) the evidence-based standard of practice to address reliability of the clinical process being addressed. The huddles also include a gap analysis document that helps assess the organization’s current state compared to its intended state.
Using latent root causes to identify system initiatives and electronic decision support
Recurring themes from event root causes are analyzed to identify those that could be prevented by EHR decision support tools or that need additional focus through a formal performance improvement project. Decision support tools may include a documentation prompt or alert; they can also include logic that helps point-of-care staff execute the clinical process. One example of such support relates to the safe administration of paralytic medications. The EHR can search the medical record for an active ventilator order or documentation before the nurse is allowed to administer a paralytic medication to a patient. Another example includes standardizing the radiology studies needed and the information communicated to the radiology technician and radiologist when an x-ray is needed in a procedural area related to a missing surgical item.
Root causes of events are queried on an ongoing basis. These reports are used to sponsor more formal process improvement initiatives related to patient safety. Such initiatives frequently focus on reducing the human-factor root causes identified in events through standardizing the processes of care and recognizing that human factors will occur (e.g., noise, distraction, fatigue, or loss of situational awareness). The focus is on designing more highly reliable clinical processes with appropriate redundancies that can withstand human factors.
Summary and future need for development
The shared learning model can be effective in rapidly communicating proximate and latent root causes of events, helping to prevent the same event from occurring in another department or organization. Events are isolated with rapid deployment of system solutions and a focus on double-loop learning for point-of-care staff (Tucker & Edmondson, 2003). This approach allows for constant refinement and improvement of care delivery through maintaining mindfulness of human factors and other latent forces that disrupt work and decision-making. The model can be used at the organization or system level as an operational model to support mindfulness and the creation of a culture of high reliability.
Ongoing opportunities with this model exist and need further study and development. Is this the best methodology to communicate with the CEO about events? What would be the most effective methodology to index safety flashes and huddles and promote their use throughout the department, organization, and system? What is the best methodology to reissue huddles and safety flashes, and how often should they be reissued? What is the best way for staff to access and use these references to assist in decision-making? (Rosness, 2009)
Jeffrey Klenklen, RN, MS-PSL, MS, CNA-BC, CPHQ, CPHRM, is a 20+ year quality management expert who specializes in helping organizations use leading practices to achieve high reliability.
References
Ahluwalia, S. C., Damberg, C. L., Silverman, M., Motala, A., & Shekelle, P. G. (2017). What defines a high-performing health care delivery system: A systematic review. The Joint Commission Journal on Quality and Patient Safety, 43(9), 450–459.
Austin, J. M., & Pronovost, P. J. (2015). “Never events” and the quest to reduce preventable harm. The Joint Commission Journal on Quality and Patient Safety, 41(6), 279–288.
Birnhack, D. J., Rosen, L. F., Williams, L., Fitzpatrick, M., Lubarsky, D. A., & Menna, J. D. (2013). A framework for patient safety: A defense nuclear industry–based high-reliability model. The Joint Commission Journal on Quality and Patient Safety, 39(5), 233–239.
Carayon, P., Wetterneck, T. B., Rivera-Rodriguez, A. J., Hundt, A. S., Hoonakker, P., Holden, R., & Gurses, A. P. (2014). Human factors system approach to healthcare quality and patient safety. Applied Ergonomics, 45(1), 14–25.
Chassin, M. R., & Loeb, J. M. (2013). High-reliability health care: Getting there from here. The Milbanks Quarterly, 91(3), 459–490.
Haig, K. M., Sutton, S., & Whittington, J. (2006). SBAR: A shared mental model for improving communication between clinicians. The Joint Commission Journal on Quality and Patient Safety, 32(3), 167–175.
Institute of Medicine (2000). To err is human: Building a safer healthcare system. Washington, DC: National Academy Press.
The Joint Commission. (n.d.). Sentinel event policy and procedures. Retrieved from www.jointcommission.org/sentinel_event_policy_and_procedures
Karl, R., & Karl, M. C. (2012). Adverse events: Root causes and latent factors. Surgery Clinic of North America, 92(1), 89–100.
Klenklen, J. (2017). Learning from actual and near miss events using debriefing methodology. The Director: Official Journal of the National Association of Directors of Nursing Administration in Long Term Care, 25(1), 19–23.
Roberts, K. H. (1990). Some characteristics of one type of high reliability organization. Organizational Science, 1(2), 160–176.
Rosness, R. (2009). A contingency model of decision-making involving risk of accidental loss. Safety Science, 47(6), 807–812.
Tucker, A. L., & Edmondson, A. C. (2003). Why hospitals don’t learn from failures: Organizational and psychological dynamics that inhibit system change. California Management Review, 45(2), 55–72.
